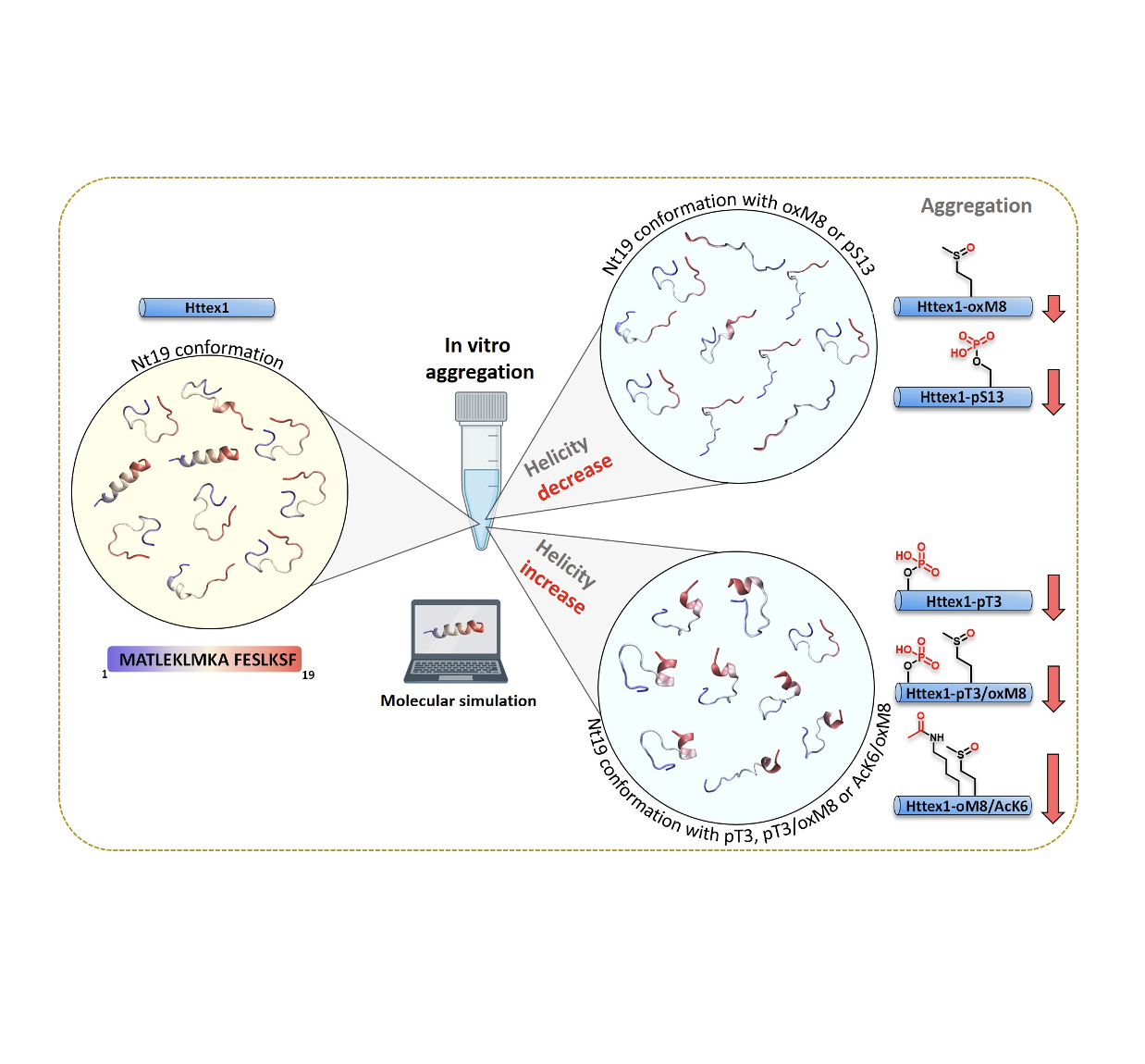
Impact of PTMs on HTT protein structure, aggregation, and membrane interaction
Huntington’s disease (HD) is a neurodegenerative disorder caused by an expanded polyglutamine stretch near the N-terminus of the huntingtin (HTT) protein, rendering the protein more prone to aggregate. We combined biophysical experiments and molecular dynamics simulation to study HTT, whose PTMs within the first 17 amino acids (Nt17) influence the conformation, membrane interaction, aggregation and toxicity.
We show that oxidation at M8 (oxM8) delays but does not inhibit the aggregation, yet the presence of both oxM8 and acetylation at K6 (AcK6) drastically inhibit mutant HTT exon 1 (mHttex1) fibrillization. 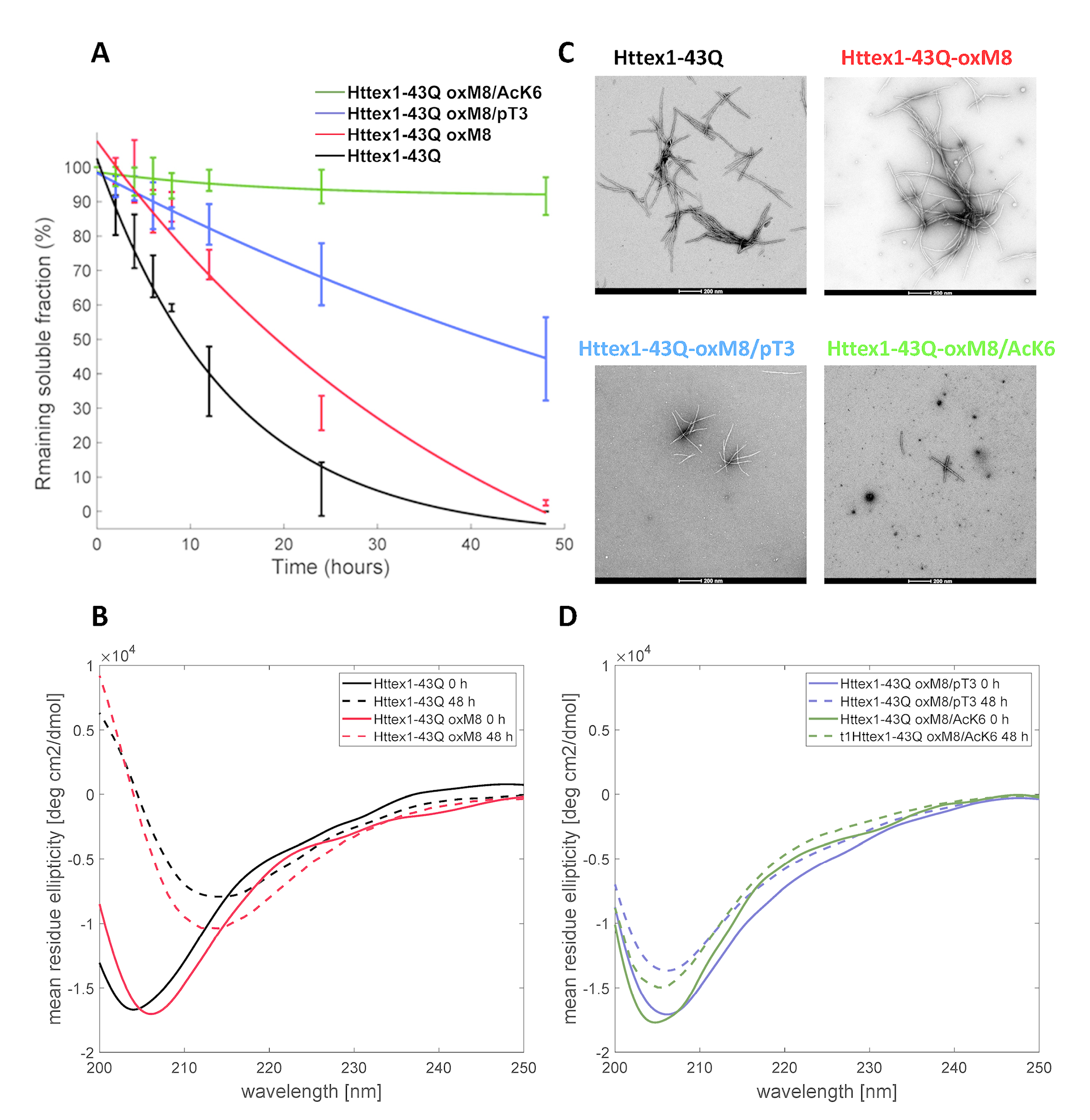
Dihedral angel PCA analysis show that PTMs that lower the mHttex1 aggregation rate result in an increased population of a short N-terminal helix in Nt17 or decreased abundance of other helical forms. 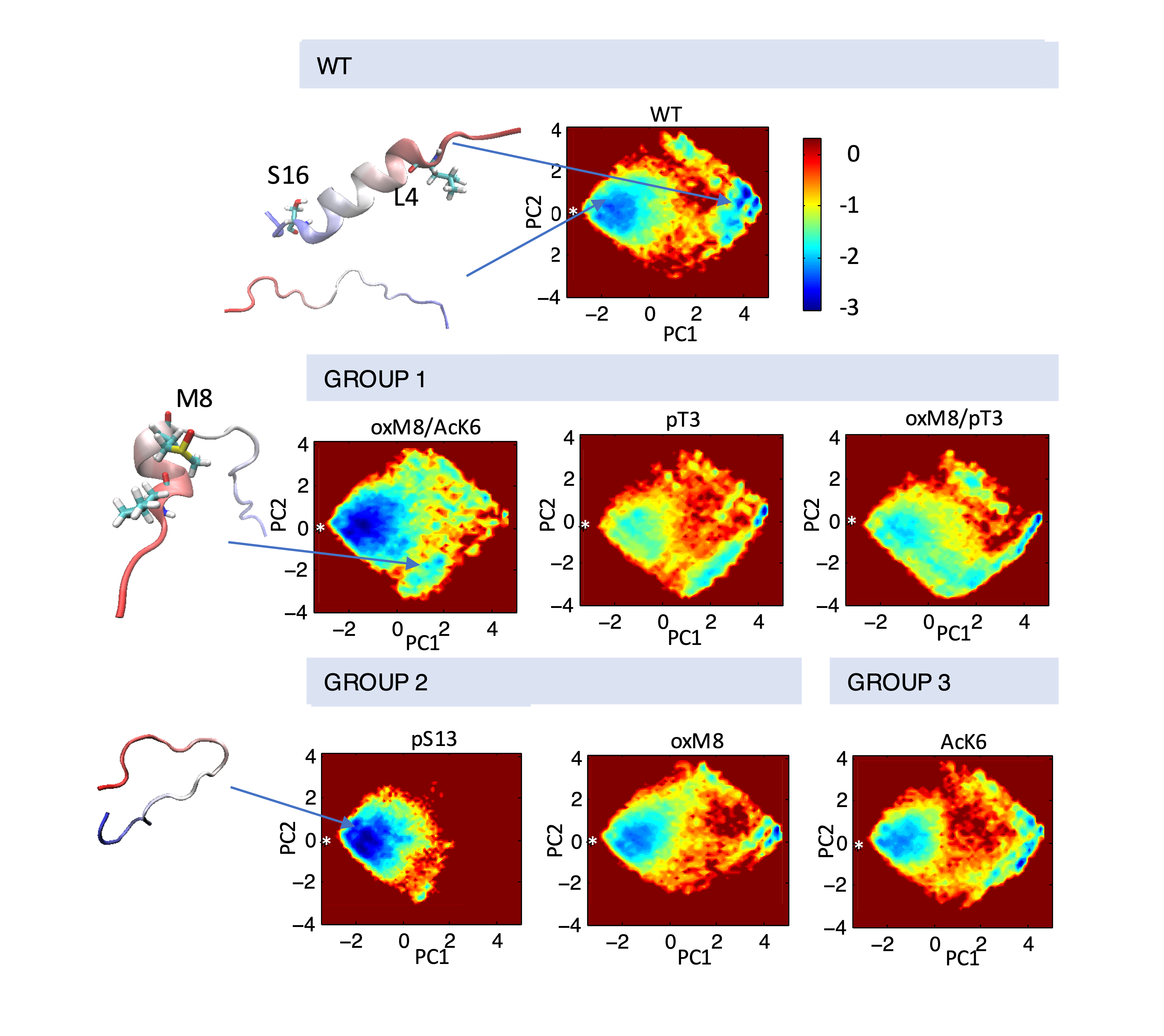
This underscores the importance of N-terminal versus C-terminal helical conformation abundance and their influence on mHttex1 aggregation, challenging the assumed correlation between overall Nt17 helicity and aggregation. Our results offer new structural perspectives on the differential effects of PTM crosstalks in regulating mHttex1 aggregation.
Then, we extended our study to the impact of PTMs on Nt17 helicity in the presence of various lipid membranes.The profound influence of membrane lipids in HD is highlighted by the impaired lipid metabolism of gangliosides in HD patients’ fibroblasts and disrupted ganglioside levels in HD patients’ caudate samples. Further, changes in the sphingomyelin fatty acid composition have been observed in cerebral white matter in HD patients
We used a combination of biophysical studies and molecular simulations to investigate the impact of PTMs on the helicity of Nt17 in the presence of various lipid membranes.
Our CD experiments for differentially modified Nt17/19 in the presence of 17 different types of membrane shows various helicity changes for each combination. 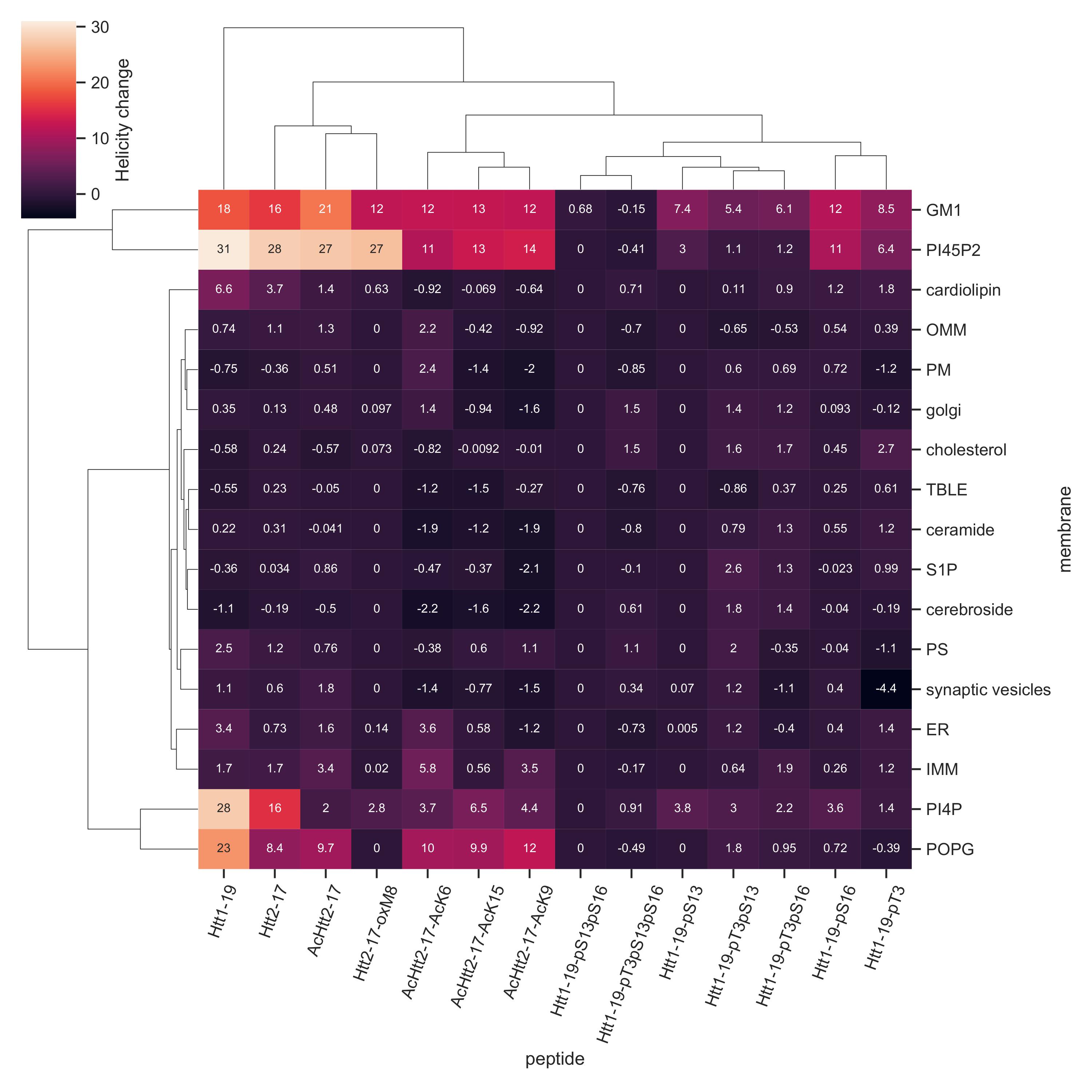
We demonstrate that anionic lipids such as PI4P, PI(4,5)P2, and GM1 enhance the helical structure of unmodified Nt17. Single acetylation at K6, K9, or K15 attenuates such effects, whereas tri-acetylation abolishes Nt17 membrane interaction. Single phosphorylation at S13 and S16 decreases but does not abolish the POPG and PIP2-induced helicity, while dual phosphorylation diminishes Nt17 helicity, regardless of lipid composition. pT3 reduces membrane interactions. oM8 variably affects membrane-induced helicity, highlighting a lipid-dependent modulation of Nt17 structure.
For simulations, we first performed clustering with CLoNe based on backbone dihedral angles for the 13 µs simulation for Nt19 corresponding in-solution simulations. The major conformations were then used as starting conformations for 5 µs simulation in the presence of a membrane. 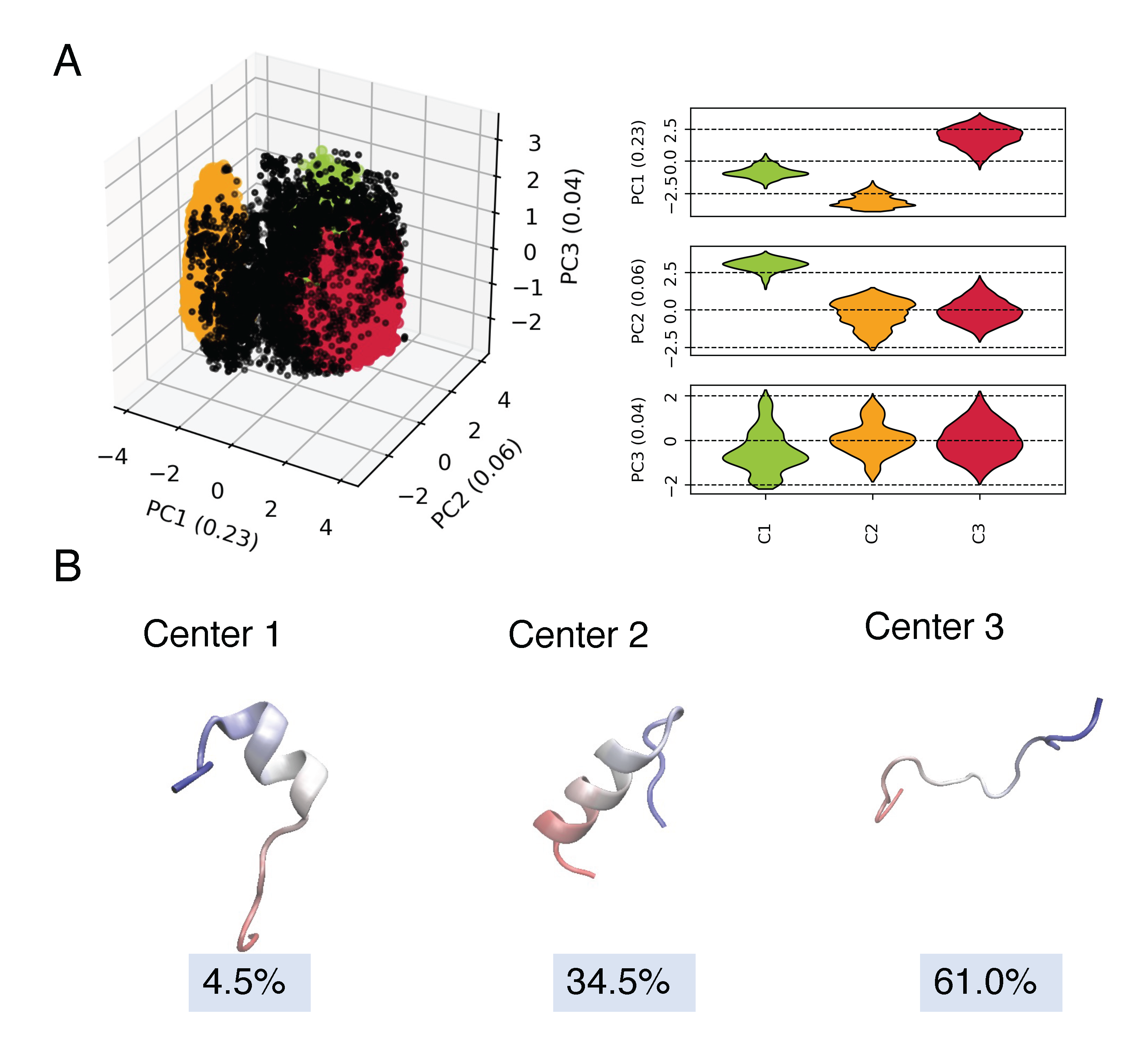
Simulations show the following:
Certain in-solution conformations were particularly stabilized by the membrane. 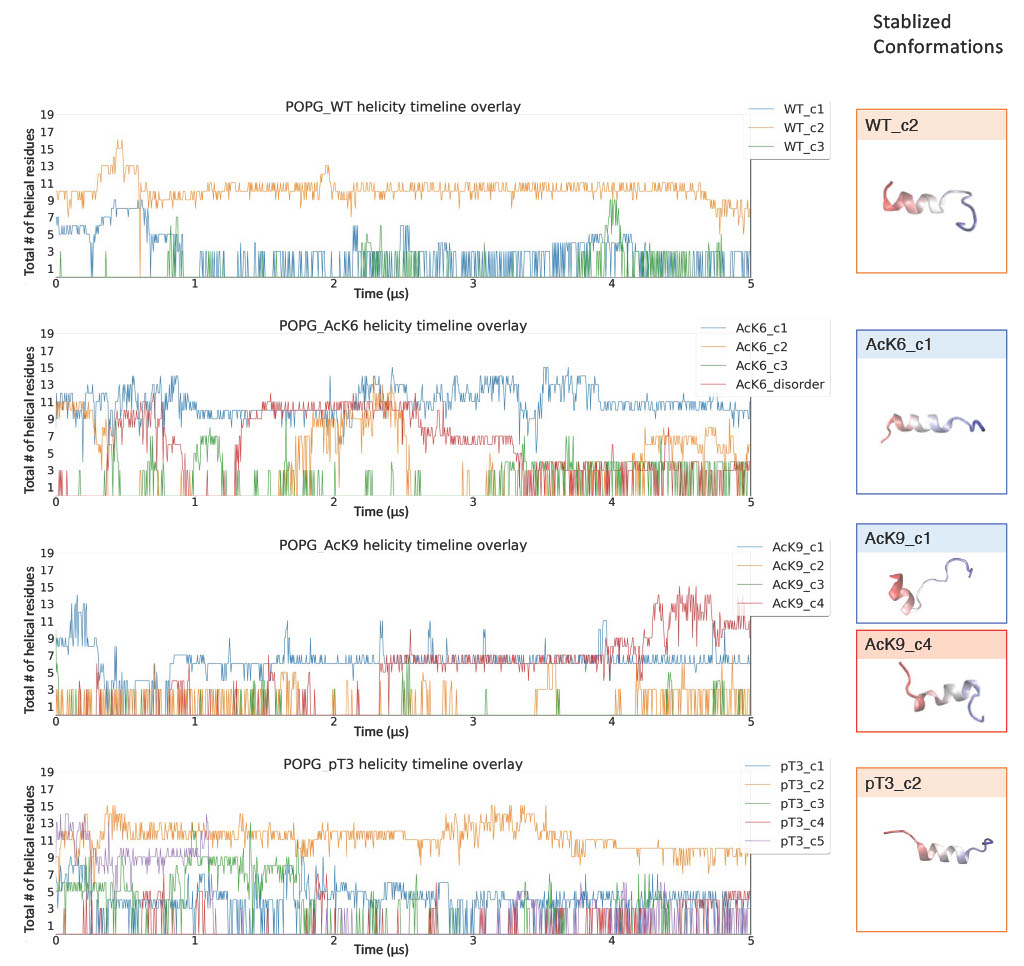
Phosphorylation at T3 disrupted the stabilization of Nt19 helical conformation by the membrane. 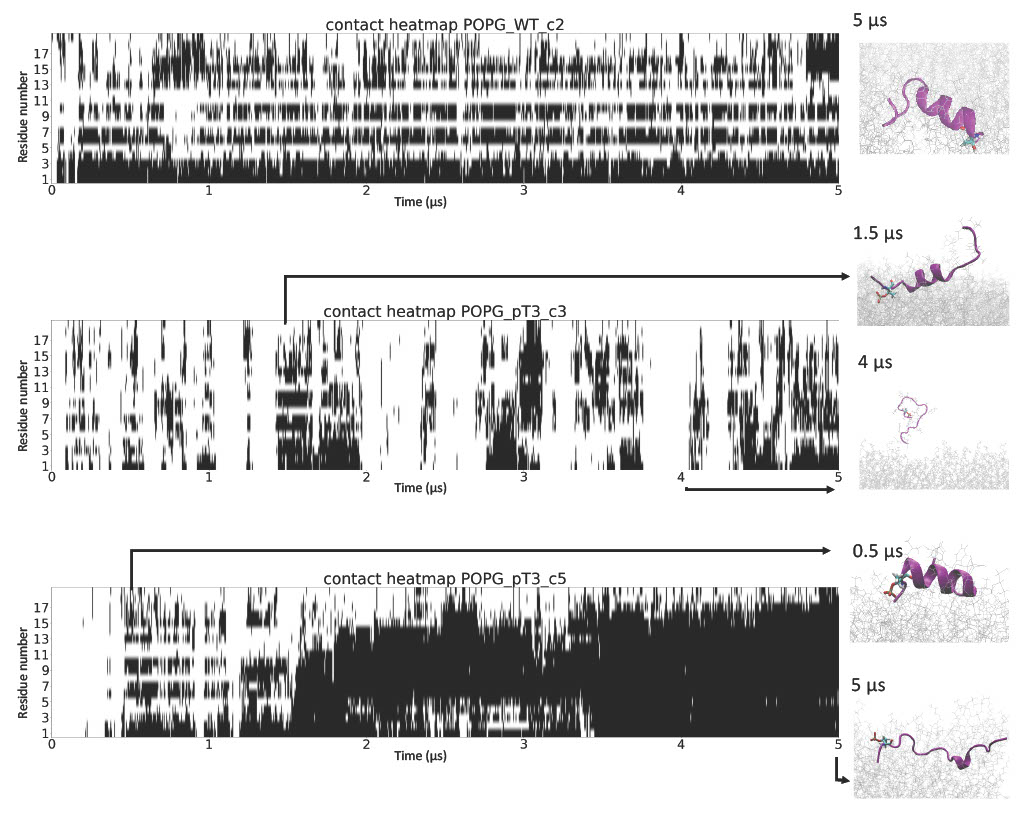
Lysine residues are essential for membrane interaction. 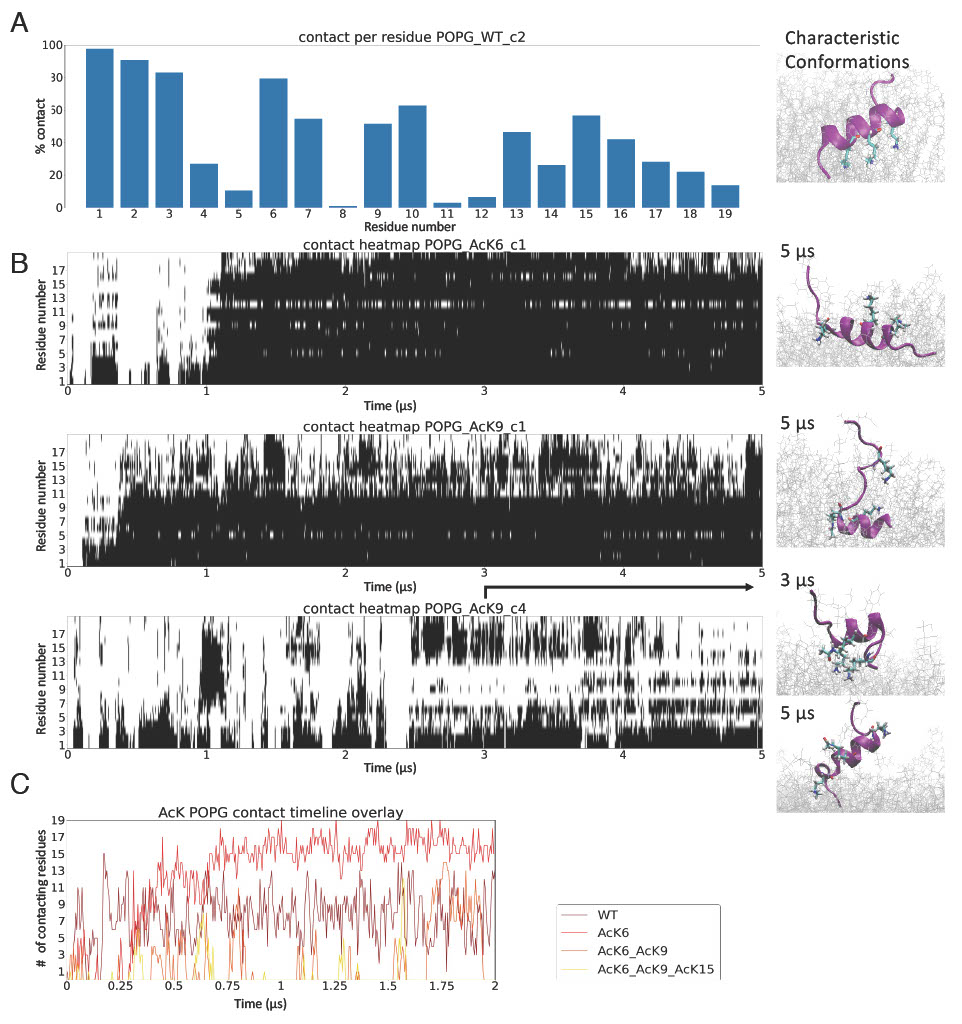
Enrichment of SV mimetics with PI45P2 resulted in increased stabilization of helical conformations. 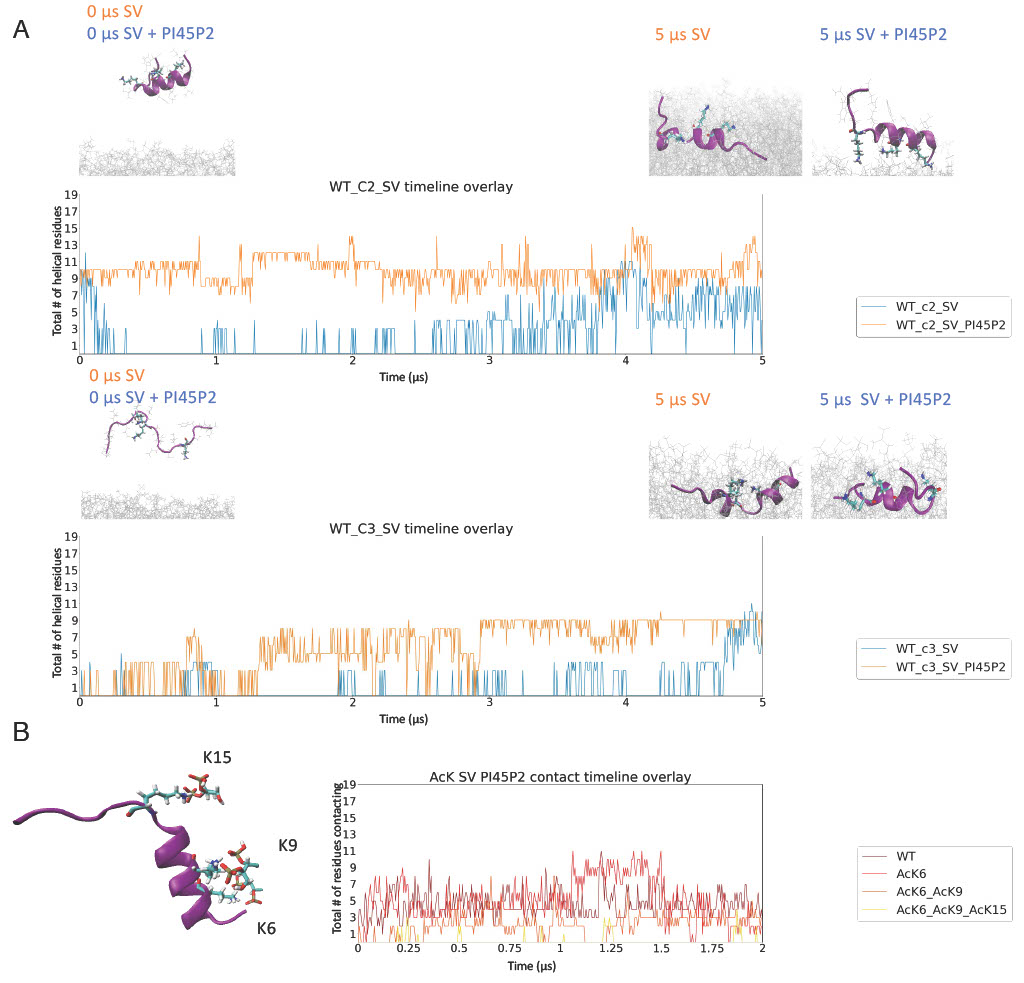
PI45P2 has a particularly high interaction with unmodified Nt19 compared to other lipids in plasma membrane. 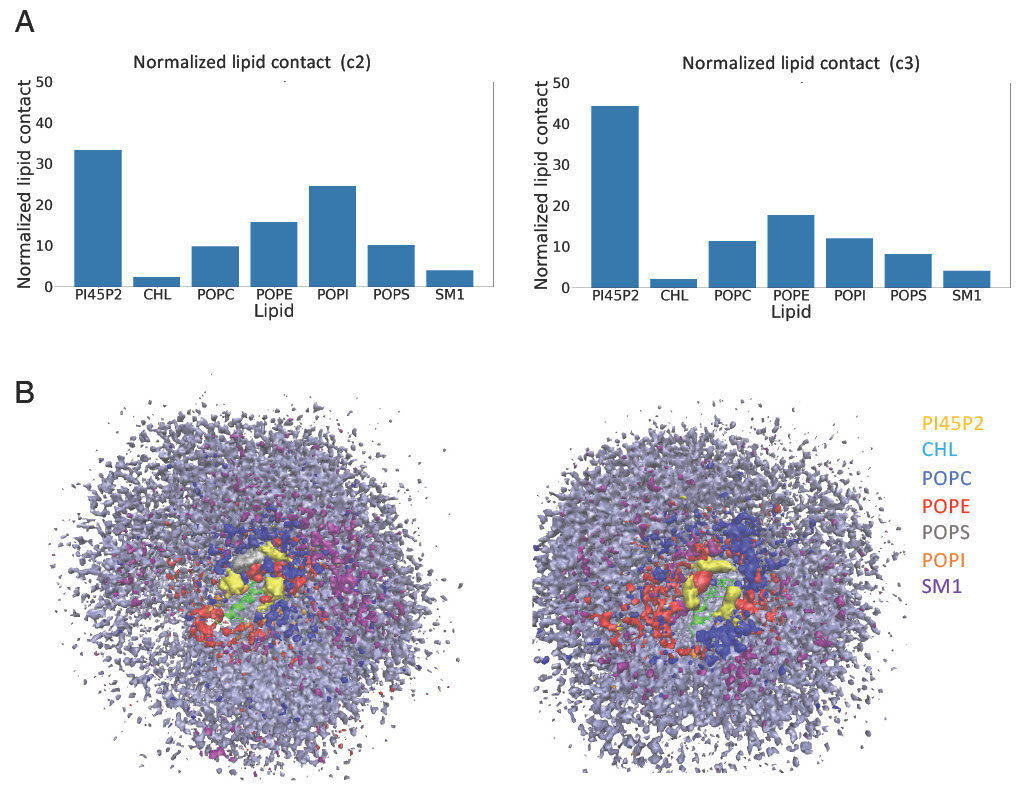
Our research uncovers the distinct effects of PTM crosstalks on membrane interaction and conformation of Nt17, providing new understanding of the complex relationship between Nt17 structure, PTMs and membrane binding.
https://www.frontiersin.org/journals/molecular-biosciences/articles/10.3389/fmolb.2021.686086/full https://pubs.acs.org/doi/full/10.1021/acschemneuro.4c00091